Diaryl Sulfone Synthesis
A three-component palladium-catalyzed coupling of aryl lithium compounds; sulfur dioxide; and aryl, heteroaryl, and alkenyl (pseudo)halides yields a diverse library of sulfones


A three-component palladium-catalyzed coupling of aryl lithium compounds; sulfur dioxide; and aryl, heteroaryl, and alkenyl (pseudo)halides yields a diverse library of sulfones
Chem. Soc. Rev., 2014, Advance Article
DOI: 10.1039/C3CS60197H, Review Article

The pentasaccharide moiety of thornasteroside A, the first asterosaponin isolated from starfish, has been synthesized for the first time

A new synthetic route to hainanolidol and harringtonolide offers flexibility to access other members of Cephalotaxus norditerpenes

A new method has been developed to selectively hydrogenate imine bonds by using an iridium complex

Benzannulation of phthalic anhydrides with alkynes to polyfunctionalized naphthalenes and phenanthrenes was confirmed to be straightforward using a palladium catalytic system. Sequential liberation of CO2 and CO occurred via oxidative decomposition of anhydride. In the case of 1,8-naphthalenedicarboxylic anhydrides, both aryls were encompassed in the annulation reaction to afford acenaphthylenes.
allow slideshare to load………….
New Drug Discovery from natural products

A cough is a response of the physique to irritants affecting the respiratory system. It is normally outlined as the unexpected act of expelling air from the lungs with a sharp sound. It can be a deliberate or involuntary act. Generally, it is the body&rsquos way of expelling bacteria and should not be suppressed
There are 2 significant varieties of coughs:
Aside from these two broad types a cough may possibly also be described as:
For the remedy of coughs triggered by an ailment or a viral, it is proposed not to use antibiotics as it can get rid of the good microorganisms, but to allow the body&rsquos immune method just take its course in working with the difficulty.
Some all-natural solutions can be followed to offer symptomatic relief whilst the an infection operates its system. These remedies may prove to be much better than the above-the-counter cough medicines obtainable at the stores, as people medicines have a tendency to suppress the cough which can consequence in a large and choked up feeling.
Honey, known for its analgesic houses helps immensely in calming a cough. It is also wealthy in bacteria and virus preventing components.
Lemon is prosperous in anti-oxidants and Vitamin C which help boost the immune system and pace up the therapeutic method, specifically in the circumstance of a cough or cold. The acidic mother nature of a lemon assists in neutralizing the phlegm that might buildup during a cough an infection. It can be used in the following techniques:
In addition to the above talked about cures utilizing lemon and honey there are a few other normal cures for cough:
For a fast recovery from any infection, it is essential to get adequate relaxation, drink lots of fluids, and steer clear of sugary and salty foods, alongside with dairy goods and meals that incorporate starch as they might aggravate mucus buildup.
In addition, keep a wholesome life style, stay away from tobacco ingestion, and use filtered masks in locations where there may possibly be heavy pollution.
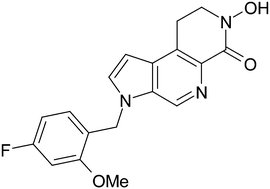
In the present article, we describe SAR studies within a series of N-hydroxy-dihydronaphthyridinone HIV integrase inhibitors that led to a candidate compound, PF-4776548, of high potency and with an excellent resistance profile. Uncertainties around the human pharmacokinetic predictions for PF-4776548 led to the compound being taken into a human microdose study to confirm its human pharmacokinetics, the results of which are described herein.